Programmed cell death (apoptosis) is a beneficial process by which biochemical processes signal the cell to alter its characteristics (morphology) and die. Apoptosis is what allows fingers and toes to separate, because the cells between the digits die. It is unlike necrosis, which is a form of traumatic cell death resulting from an injury. Apoptosis is a huge area of study in biology and medicine, because the inhibition of the apoptotic pathway can lead to a number of cancers, autoimmune diseases, inflammatory diseases, and viral infections.

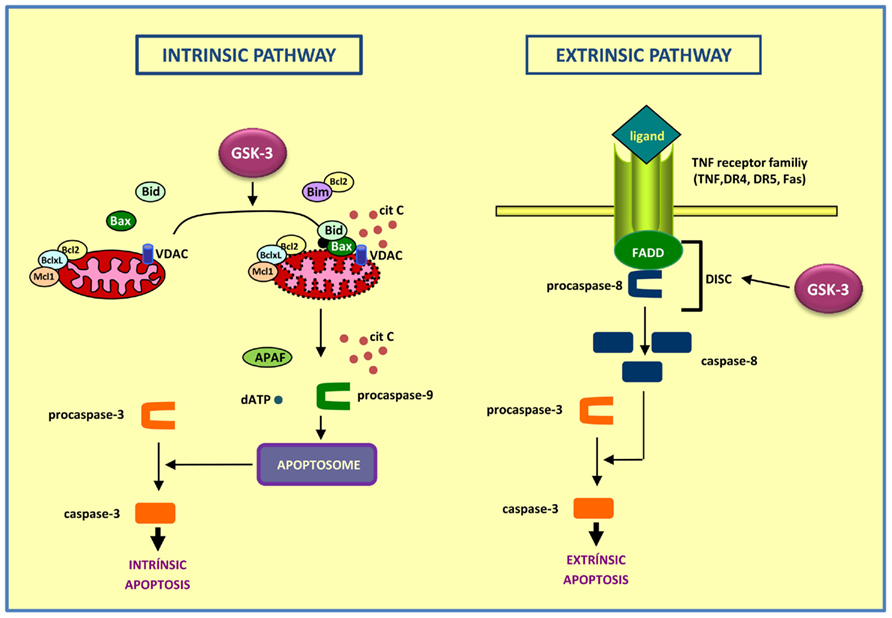
The way it works is, cells release an intracellular apoptotic signal in response to a stress. There are two ways in which this signal is received: either by affecting mitochondrial function (intrinsic), or through directly passing the signal to the apoptotic mechanisms (extrinsic). The mitochondrion is the part of the cell that is responsible for aerobic respiration and for essentially making all the energy that your cells require to survive. If signals are received that alter its function, this tells the cell that it is essentially committing suicide and triggers the apoptotic mechanism. On the other hand, passing the signal directly onto the apoptotic mechanisms, in a process called signal transduction - which is basically like a relay race through the cell passing on a signal from one protein to another - by-passes the mitochondrial step. Either way, the end-result is the same, and each way can be reversed if cells no longer need to die.
 (source)
(source)
Apoptosis and autophagy are closely linked. Autophagy comes from the Greek "self" and "to eat", so that's pretty self-explanatory. The process of autophagy is when a cell begins to degrade unnecessary or dysfunctional cellular components, which can ensure survival by maintaining cellular energy levels during starvation stress. Sometimes (and this is gross), animals use autophagy to avoid starvation by eating their own limbs.
On the cellular level, autophagy and apoptosis are thought to be opposite pathways, and there are distinct mechanisms through which apoptosis can inhibit autophagy. But the process through which autophagy protects cells against apoptosis is not well understood. Well, a group out of the U.S. recently found a way in which autophagy can control apoptosis and determine cell-fate decisions. This study is being published in Cell in the next few days, but is already available online and is open access which makes me unbelievably happy!
Typically, the point-of-no-return in apoptosis is the point in which mitochondria are broken down, called Mitochondiral Outer Membrane Permeabilization, or MOMP (those cell biologists love their acronyms, so sometimes it's hard to keep up). This group, led by Jacqueline Thorburn, found that autophagy can control apoptosis by regulating the timing of MOMP. They treated HeLa cells with a tumor necrosis factor-related apoptosis inducing ligand (TRAIL), and monitored MOMP using time lapse microscopy, and cells died within 5 minutes. But when autophagy was induced, MOMP still happened, but it was delayed. That's because autophagy regulates the levels of a protein called PUMA (p53 upregulated modulator of apoptosis), which is key in MOMP.
Once apoptosis gets started, cells contract and die fairly quickly. But when autophagy was induced in the cells, cell contraction after MOMP was delayed. When this happened, some of the cells were able to recover after MOMP and display a normal morphology, which is surprising. The more autophagy and PUMA degradation present in the cells, the better the cells were able to recover from MOMP. The use of time lapse microscopy also means that there are videos of this, which the authors included in their supplemental information - and isolated frames are included in the text of the article as figures.
This is time lapse microscopy showing inefficient MOMP and eventual cell recovery and division in cells that were not expressing PUMA. I've also included the relevant part of the in-text figure, so that you can have an idea of what you're looking at. These all come from the paper, so as usual, if you're going to be using any of this, please cite properly.
So what's happening here is that in the first few frames, the mitochondria are undergoing MOMP, but then high autophagy allows the cell to engulf the released mitochondrial proteins, which keeps the rest of apoptosis from occuring, thus allowing the cells to recover and ultimately divide. The authors speculate that this may be the process that allows cancer cells to recover and rescue themselves under stress from chemotherapy, with the implication being that if we can find a way to inhibit autophagy, then maybe we can increase the chance of cancer cell death during treatment.

Okay I have a question. My mom has a tumor inside her but all the cancer cells inside it are dead. If this is what's happening, is this the reason why the tumor is still intact? The cells are still there, but dead?
ReplyDelete